Blog
Gipr signaling opens the brain gate to GLP-1 slimming therapy
In the last study published in the journal A group of scientists has checked whether insulinotropic polypeptide receptor signaling (GIPR) in oligodendrocytes (OLS) increases brain access and weight loss dependent on glucose. effectiveness Agonists of the Peptic-1 Glucagon (GLP-1R) receptor.
Background
One in eight adults live with obesity, and many now use incretin drugs that can reduce weight by over 20%. Incretins work through GIPR and GLP-1R, but why their connection helps to remain unclear. Median Eminence (ME), an interface in which blood signals meet neurons can be a gate.
OLS, long known for making myelin, also rebuild this gate in response to your diet. Explanation whether insulinotropic (GIP) depending on glucose (GIP) in OLS increases the brain input, and the effects of peptide-1 therapy similar to glucagon (GLP-1) can conduct stronger treatment; Further research is needed.
About the study
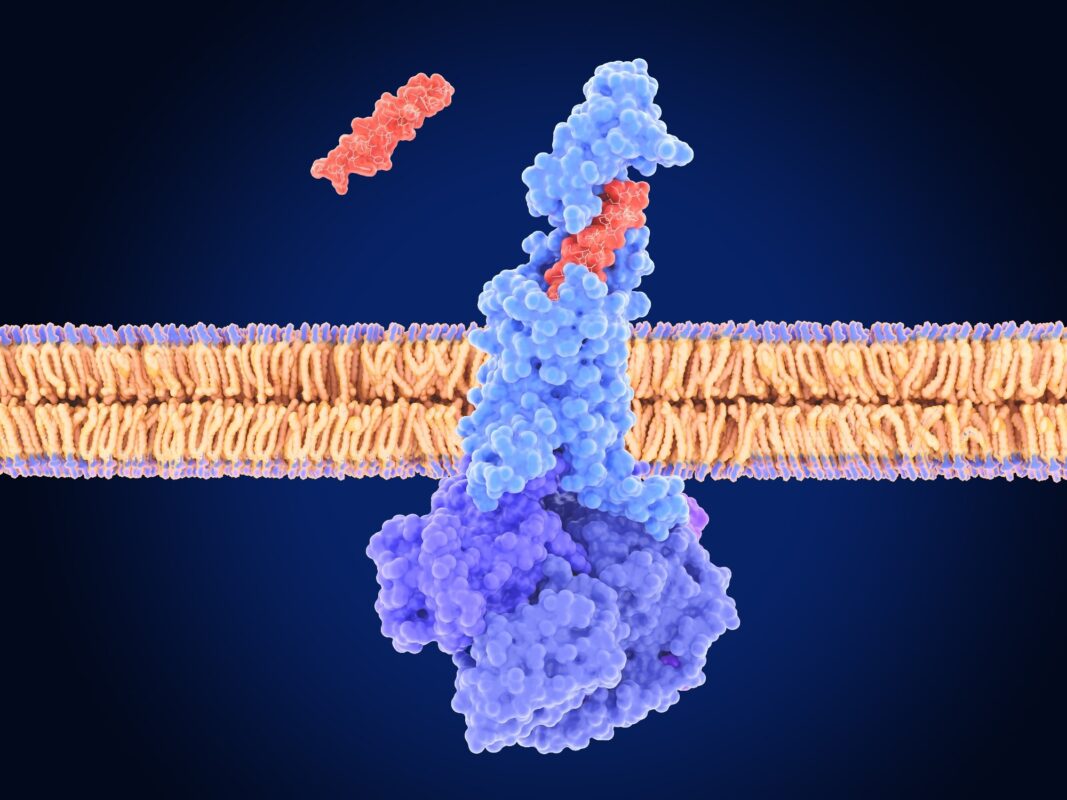
Scientists used adult mice to see if GIPR signaling in OLS shapes brain access and the effectiveness of GLP-1R agonists. They generated inductive OL knockout by exceeding the proteolipid protein 1-CRE receptor-t2 receptor (PLP1-Creert2) with Floxed Gipr mice, launched recombination with tamoxifen on postpartum day 60 and induced obesity with 60% high-fat diet. The long-acting GIPR agonist (Lagipra) and the long-acting GLP-1R agonist (Lagglp-1ra; Liraglutide) were served alone or together.
To map the introduction of drugs, the short-acting GLP-1R marked agonist IR800 (IR800-Exendin-4) was injected; The brains have been cleaned and illustrated with light microscopy. OLODENDENDROGEnesis and myelinization were quantitatively determined by fluorescent hybridization in situ and immunological coloring for basic myelin protein, amplified sequence of breast cancer 1 and bone morphogenetic protein 4, with pulsed yoke of 5-ethyna-2′-deoxyuridine.
Vascular permeability was assessed by vascular endothelial growth factor Expression (VEGF-A), Immunoreactivity of the vascular endothelial growth factor (VEGF) and mouse antigen of endothelial cell 32 (MECA32) fenestred capillaries. Metabolic readings included energy expenditure, food intake and glucose and insulin tolerance.
Finally, the virus associated with the adeno encoding the M4 muscarin receptor designed for activation by designer drugs (HM4DI) directed the hypothalamus of the screens (AVP-CRE); Deschloroclosopine is administered to activate HM4Di and suppress these neurons during lyreglutide tests.
Research results
In ME, GIPR was enriched with mature ols, with rare expression in progenitor cells; High feeding content increased the density of OL and the number of GIPR-Dodatnich OLS in this region. This effect was not observed in the areas of white being, such as Callosum bodies, which indicates a located role.
The GIPR deletion specific to Ol reduced adult oligodendrogenesis and OL survival in ME and reduced myelin protein, while the main white patenchers showed a slight change, which indicates a role in the brain metabolic gate. Mice without OL GIPR showed reduced energy expenditure and consumption, preserved oral glucose tolerance, insulin tolerance and the shift of metabolites associated with a branched chain, in accordance with the change in the support of the substrate during obesity.
Pharmacological activation caused supplementary effects. In slim mice, the long -acting Gipr agonist increased lines and myelin cells. In an induced diet (DIO) obesity, the same agonist increased the new OL production and restored rotation, while raising vascular access signals: VEGF-A transcripts and immunoreactivity Vegf increased, and the capillaries marked by MECA32 have become Denter, which indicates increased vascular permeability.
Initial processing of obese mice with agonist GIPR increased the download of the brain marked by 800 short GLP-1R agonist in the European Championships and the neighboring arc of the hypothalamus (ARH), which indicates increased access at the ME-ARH border. Most importantly, OL GIPR required an increase in this growth; He was not after removal of OL GIPR.
Effectiveness was reflected in the entrance, just like in wild-type mice, the long-acting GLP-1R agonism reduced food intake and body weight, as well as cooperation with the GIPR agonist strengthened both results; In knockouts OL GIPR, the GIPR agonist does not allow weight loss or powered anorexia GLP-1R, which indicates that OL GIPR signaling is required for full synergy.
Imaging showed that the peripherally administered short-acting GLP-1R agents gathered along the myelin-bunch of ME axons, colloquializing the basic myelin protein; The disclosure of the innovative mechanism: The peripherally administered GLP-1R agonists gain access to the brain through myelinated AVP axons in the European Championships, bypassing the blood-brain barrier (BBB).
Super-dispersion microscopy located GLP-1R on AVP axons and in nodes marked with contact protein (CasPR). Finally, the chemogenetic silencing of AVP PVH Deschloroklozapine neurons prevented hypophagia induced with liraglutide and weight loss, which shows that these neurons are necessary to operate the load on a systemic drug.
Conclusions
To sum up, this study combines incretin pharmacology with a concrete brain input mechanism: GIPR signaling in ME OLS increases vascular permeability by VEGF induction and increased capillary fenestration and enables GLP-1R agonists to achieve AVP axons with an appetite regulator.
The requirement of this trail can help explain why co-authors of GIPR/GLP-1R show greater efficiency than individual means and constitute mechanistic foundations of their increased clinical efficiency. Clinically, the mechanism helps to interpret the power of co-donated for obesity and type 2 diabetes as well as biomarkers points such as VEGF induction or ME Access imaging to conduct dosage or combinations, limiting side effects.
However, the authors notice a few restrictions: the OL GIPR knockout model reached only partial removal; Experiments were primarily assessed by liraglutide, not other GLP-1R agonists; And behavioral results, although informative, were not exhausting. These reservations develop conclusions and emphasize the need for further research to confirm generalization and clinical significance.
Why Zepbound (Tirirpatide) leads to greater weight loss than ozempic (Semglutide)?
The double receptor (GLP-1+ GIP) has a more pronounced operation in the brainhttps://t.co/1aldifds2o pic.twitter.com/ws81pf5cxf– Eric Topol (@ericictopol) August 13, 2025
Reference to the journal:
- Hansford, R., Buller, S., Tsang, Ah, Benoit, S., Roberts, AG, Erskine, E., Brown, T., Pirro, V., Reimann, F., Harada, N., Inagaki, N., Sams, RJ, Broichhagen, J., Hodson, DJ, Adrianssens, A. Bloet, C. (2025). Glucose dependent signaling of insulinotropic polypeptide receptor in oligodendrocytes increases the effect of the mass of agonism GLP-1R. . Doi: 10.1016/J. CMET.2025.07.009, https://www.cell.com/cell-metolim/fulltext/s1550-4131(25)00355-9